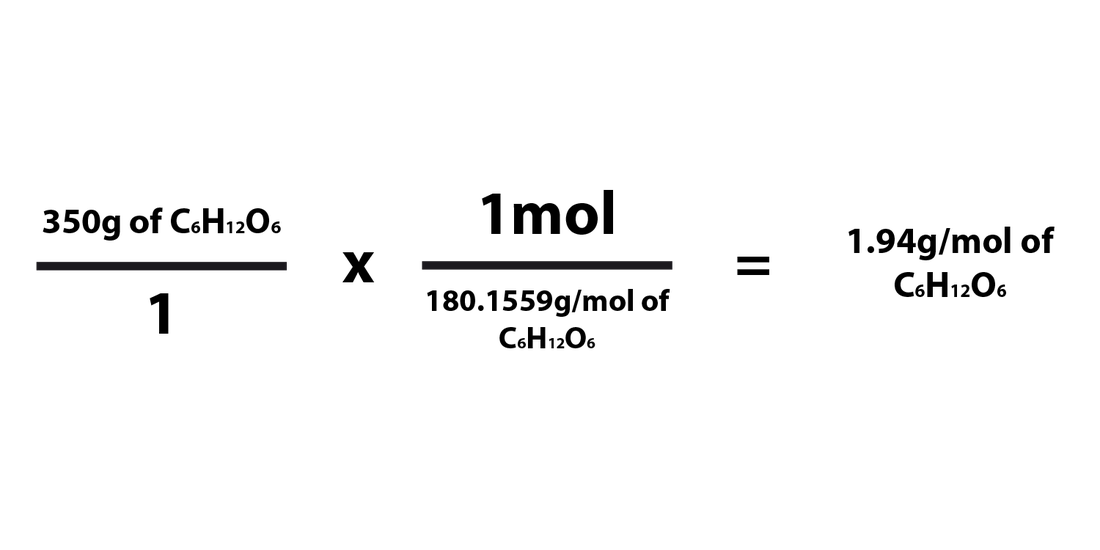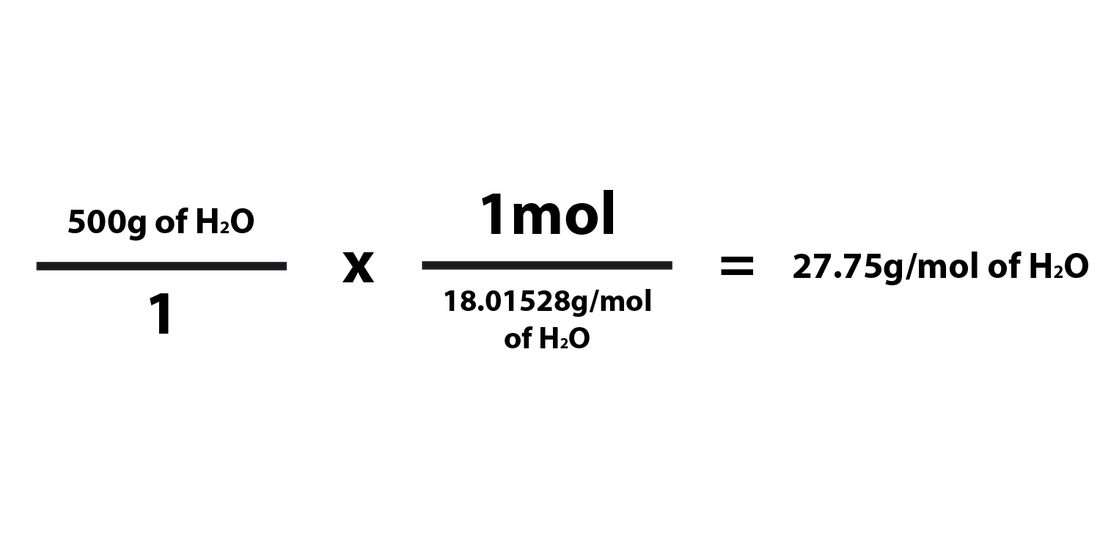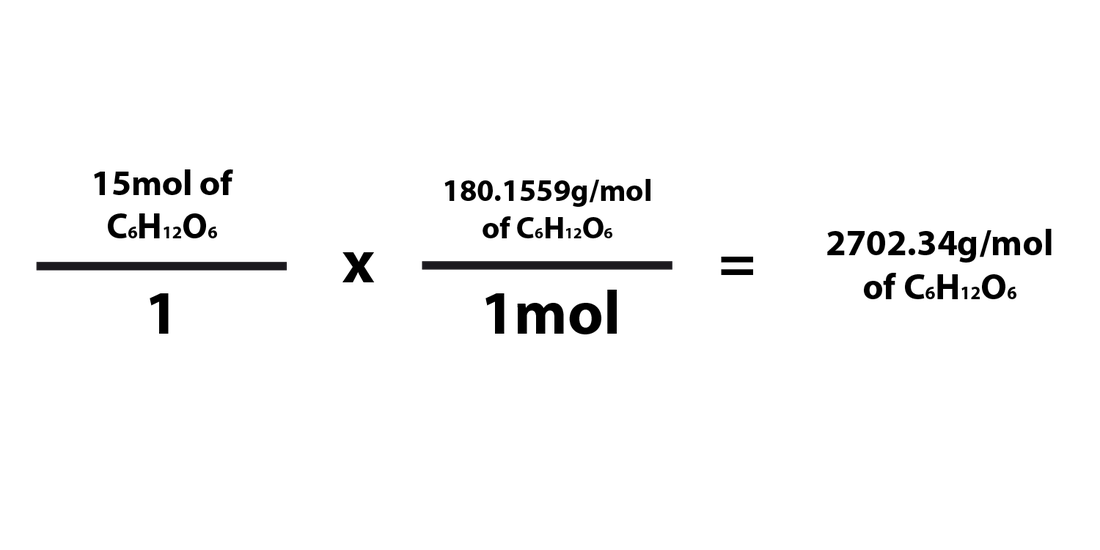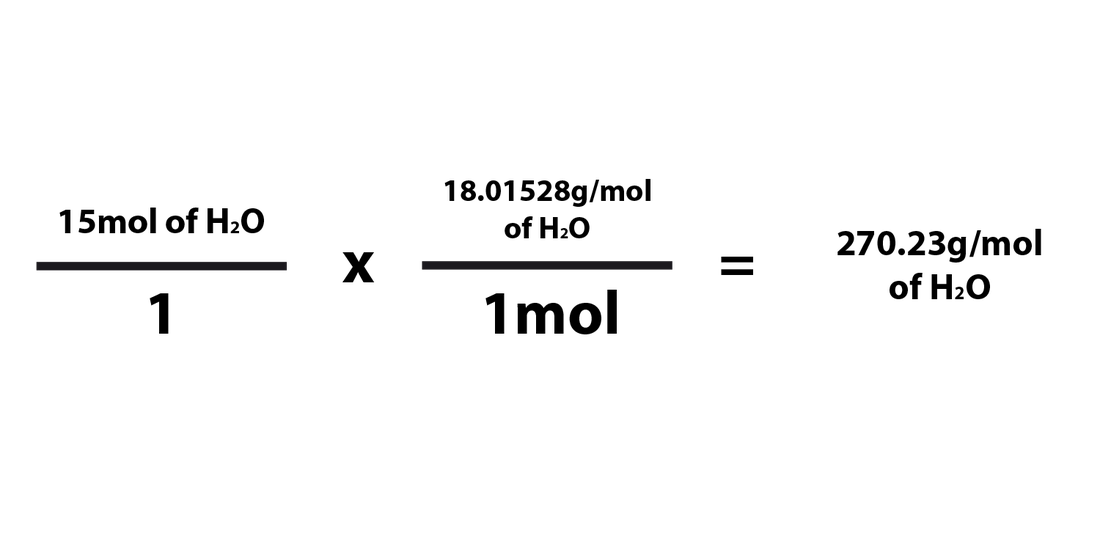Converting Grams & MolesIf you know the number of grams of an element or the number of grams of a compound, you can convert it to the number of moles of that element. Conversely, if you know the number of moles in an element, you can convert it to the number of grams of that element.
|
Converting Grams ---> Moles
When converting the number of grams of a compound/element into moles, you'd just need to multiply the number of grams by the molar mass of that compound. The formula for converting from grams to moles is as follows:
When going from grams to moles, 1mol must be in the numerator and the compound's molar mass must be in the denominator. It has to be multiplied in this way in order for the grams to cancel each other out and be replaced by the unit "mol." This works because you'd essentially be dividing the # of grams of the element/compound by the molar mass of the compound. The "mol" part of the fraction must always be "1mol" because the molar mass is written in grams per mole (g/mol).
Grams ---> Moles Examples
Converting Moles ---> Grams
When converting the number of moles of a compound/element into grams, you'd just need to multiply the number of moles by the molar mass of that compound. However, the fraction for the molar mass will have the molar mass of the compound in the numerator and "1mol" in the denominator.. The formula for converting from moles to grams is as follows:
When going from moles to grams, 1mol must be in the denominator and the compound's molar mass must be in the numerator. It has to be multiplied in this way in order for the moles to cancel each other out and be replaced by the unit "grams." You'd essentially be multiplying the # of grams of the element/compound by the molar mass of the compound in order to find the number of grams of the element/compound. As explained above, the "mol" part of the fraction must always be "1mol" because the molar mass is written in grams per mole (g/mol).